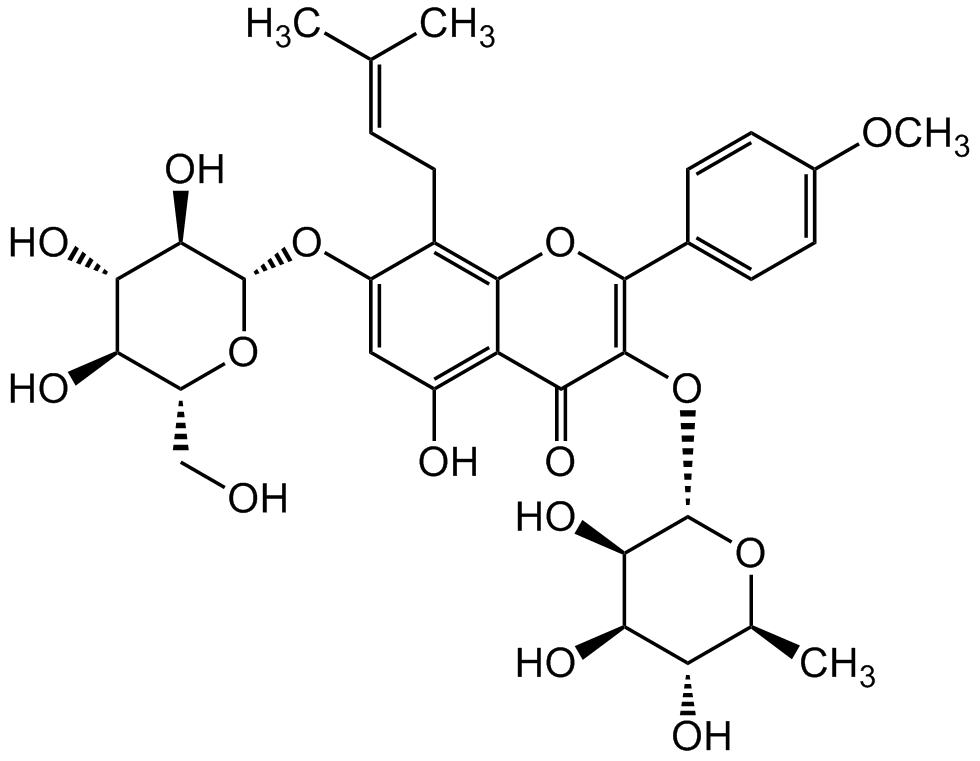
Icariin
Product Code:
CDX-I0172
CDX-I0172
Host Type:
Plant
Plant
Regulatory Status:
RUO
RUO
Shipping:
AMBIENT
AMBIENT
Storage:
Short Term: +20°C Long Term: -20°C
Short Term: +20°C Long Term: -20°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-I0172-M100 | 100 mg | £84.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Alternate Names/Synonyms:
4'-O-Methyl-8-gamma,gamma-dimethylallylkaempferol-3-rhamnoside-7-glucoside; Leariline
Appearance:
Light yellow fine powder.
CAS:
489-32-7
EClass:
32160000
Form (Short):
solid
Handling Advice:
Protect from light and oxygen.
InChi:
InChI=1S/C33H40O15/c1-13(2)5-10-17-19(45-33-28(42)26(40)23(37)20(12-34)46-33)11-18(35)21-24(38)31(48-32-27(41)25(39)22(36)14(3)44-32)29(47-30(17)21)15-6-8-16(43-4)9-7-15/h5-9,11,14,20,22-23,25-28,32-37,39-42H,10,12H2,1-4H3/t14-,20+,22-,23+,25+,26-,27+,28+,32-,33+/m0/s1
InChiKey:
TZJALUIVHRYQQB-XLRXWWTNSA-N
Long Description:
Chemical. CAS: 489-32-7. Formula: C33H40O15. MW: 676.668. Icariin, the active component of the Chinese medicinal plant E. brevicornum. It has been shown to have anti-inflammatory, anticancer, antioxidant, osteoprotective, neuroprotective and cardioprotective activity. It has been shown to enhance bone formation and bone regeneration. Icariin may be beneficial to Alzheimer's disease by reducing the production of extracellular amyloid plaques and intracellular neurofibrillary tangles and inhibiting phosphodiesterase-5 activity. The anti-cancer activity is related to various mechanisms such as apoptosis and autophagy induction, cell cycle modulation, antiangiogenesis and antimetastasis, modulating multidrug resistance and immunomodulation. Icariin interacts with several pathways, like PDE, TGF-beta, MAPK, PPAR, NOS, IGF, Sirtuin and others. Icariin has been shown to inhibit the NF-kappa activation pathway and NLRP3 inflammasome.
MDL:
MFCD00210516
Molecular Formula:
C33H40O15
Molecular Weight:
676.668
Package Type:
Vial
Product Description:
Icariin, the active component of the Chinese medicinal plant E. brevicornum. It has been shown to have anti-inflammatory, anticancer, antioxidant, osteoprotective, neuroprotective and cardioprotective activity. It has been shown to enhance bone formation and bone regeneration. Icariin may be beneficial to Alzheimer's disease by reducing the production of extracellular amyloid plaques and intracellular neurofibrillary tangles and inhibiting phosphodiesterase-5 activity. The anti-cancer activity is related to various mechanisms such as apoptosis and autophagy induction, cell cycle modulation, antiangiogenesis and antimetastasis, modulating multidrug resistance and immunomodulation. Icariin interacts with several pathways, like PDE, TGF-beta, MAPK, PPAR, NOS, IGF, Sirtuin and others. Icariin has been shown to inhibit the NF-kappa activation pathway and NLRP3 inflammasome.
Purity:
>97% (HPLC)
SMILES:
O=C1C2=C(O)C=C(O[C@H]3[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O3)C(C/C=C(C)/C)=C2OC(C4=CC=C(OC)C=C4)=C1O[C@H]5[C@H](O)[C@H](O)[C@@H](O)[C@H](C)O5
Solubility Chemicals:
Soluble in DMSO (20mg/ml) or DMF (20mg/ml).
Source / Host:
Plant
Transportation:
Non-hazardous
UNSPSC Category:
Natural Products/Extracts
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at -20°C.
Documents
References
(1) L.G. Ming, et al.; J. Cell Physiol. 228, 513 (2013) (Review) | (2) J.K. Schluesener & H. Schluesener; Mol. Nutr. Food Res. 58, 49 (2014) (Review) | (3) H.L. Tan, et al.; Front. Pharmacol. 7, 191 (2016 (Review) | (4) R. Shen & J.H. Wang; Am. J. Clin. Exp. Immunol. 7, 50 (2018) (Review) | (5) B. Su, et al.; Life Sci. 208, 26 (2018) | (6) J. Jin, et al.; Eur. J. Pharmacol. 842, 20 (2019) (Review) | (7) C. Angeloni, et al.; Front. Pharmacol. 10, 271 (2019) (Review) | (8) A. Yang, et al.; Stem Cells Int. 2019, 5747298 (2019) (Review)