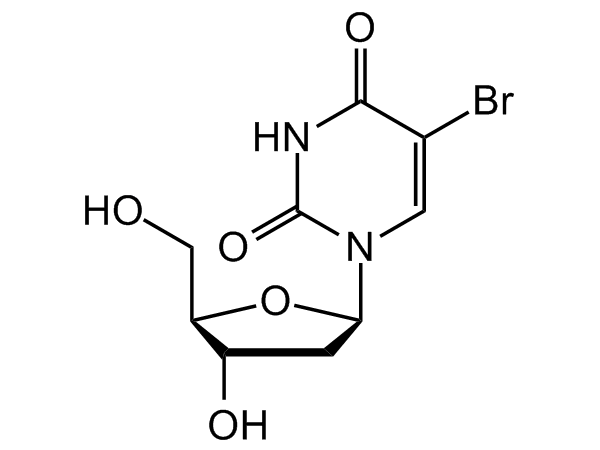
5-Bromo-2'-deoxyuridine
Product Code:
CDX-B0301
CDX-B0301
Regulatory Status:
RUO
RUO
Shipping:
AMBIENT
AMBIENT
Storage:
Short Term: +4°C, Long Term: -20°C
Short Term: +4°C, Long Term: -20°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-B0301-G001 | 1 g | £65.00 |
Quantity:
| CDX-B0301-G005 | 5 g | £218.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Alternate Names/Synonyms:
5-BrdU; 5-Bromo-1-(2-deoxy-beta-D-ribofuranosyl)uracil; 5-Bromouracil deoxyriboside; BUdR; Bromodeoxyuridine; Broxuridine; NSC 38297
Appearance:
White powder.
CAS:
59-14-3
EClass:
32160000
Form (Short):
liquid
GHS Symbol:
GHS08
Handling Advice:
Protect from light and moisture.
InChi:
InChI=1S/C9H11BrN2O5/c10-4-2-12(9(16)11-8(4)15)7-1-5(14)6(3-13)17-7/h2,5-7,13-14H,1,3H2,(H,11,15,16)/t5-,6+,7+/m0/s1
InChiKey:
WOVKYSAHUYNSMH-RRKCRQDMSA-N
Long Description:
Chemical. CAS: 59-14-3. Formula: C9H11BrN2O5. MW: 307.1. The synthetic nucleoside 5-Bromo-2'-deoxyuridine (BrdU) is a thymidine analog used to label DNA and commonly used in the detection of proliferating cells in living tissues. It can be incorporated into newly synthesized DNA in place of thymidine during the S phase of the cell cycle. Cells that were actively proliferating can then be detected by denaturing the DNA and allowing specific antibodies with fluorescent tags to target the BrdU incorporation for detection via flow cytometry or fluorescence microscopy. Binding of the antibody requires denaturation of the DNA, usually by exposing the cells to acid or heat. BrdU is also used as a mutagen in genetic research. BrdU stimulates cellular differentiation and maturation in leukemia cell lines, while it inhibits differentiation of friend erythroleukemia cells. BrdU replaces Oct-4 in transcription factor-mediated reprogramming of somatic cells and can be used in small molecule cocktail to generate ciPSCs.
MDL:
MFCD00006529
Molecular Formula:
C9H11BrN2O5
Molecular Weight:
307.1
Package Type:
Vial
Precautions:
P201-P281-P308 + P313
Product Description:
The synthetic nucleoside 5-Bromo-2'-deoxyuridine (BrdU) is a thymidine analog used to label DNA and commonly used in the detection of proliferating cells in living tissues. It can be incorporated into newly synthesized DNA in place of thymidine during the S phase of the cell cycle. Cells that were actively proliferating can then be detected by denaturing the DNA and allowing specific antibodies with fluorescent tags to target the BrdU incorporation for detection via flow cytometry or fluorescence microscopy. Binding of the antibody requires denaturation of the DNA, usually by exposing the cells to acid or heat. BrdU is also used as a mutagen in genetic research. BrdU stimulates cellular differentiation and maturation in leukemia cell lines, while it inhibits differentiation of friend erythroleukemia cells. BrdU replaces Oct-4 in transcription factor-mediated reprogramming of somatic cells and can be used in small molecule cocktail to generate ciPSCs.
Purity:
>99% (HPLC)
Signal word:
Danger
SMILES:
O[C@H]1C[C@H](N2C=C(Br)C(NC2=O)=O)O[C@@H]1CO
Solubility Chemicals:
Soluble in DMSO (30mg/ml), DMF (30mg/ml) or ethanol (25mg/ml) or water (10mg/ml).
Transportation:
Non-hazardous
UNSPSC Category:
Biochemical Reagents
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at -20°C.
Documents
References
(1) N. Kee, et al.; J. Neurosci. Methods 115, 97 (2002) | (2) R.C. Leif, et al.; Cytometry A 58, 45 (2004) (Review) | (3) P. Taupin; Brain Res. Rev. 53, 198 (2007) (Review) | (4) D.W. Stacey & M. Hitomi; Cytometry A 73, 270 (2008) | (5) B.L. Cavanagh, et al.; Molecules 16, 7980 (2011) | (6) B. Lehner, et al.; Cell Tissue Res. 345, 313 (2011) | (7) T. Konishi, et al.; J. Radiat. Res. 52, 433 (2011) | (8) J.M. Barker, et al.; PLoS One 8, e63692 (2013) | (9) Y. Long, et al.; Cell Res. 25, 1171 (2015)