Further Information
Alternate Names/Synonyms:
PKB; AKT1; PKB alpha; EC=2.7.11.1; RAC-PK-alpha; Protein Kinase B; Proto-Oncogene c-Akt; Protein Kinase B alpha; PKBG; AKT3; STK-2; PKB gamma; RAC-PK-gamma; Protein Kinase Akt-3; Protein Kinase B gamma; AKT2; PKB beta; RAC-PK-beta; Protein
1 mg/ml
32160000
liquid
Liquid. 50% Glycerol/PBS with 1% BSA and 0.09% sodium azide.
Avoid freeze/thaw cycles.
A peptide corresponding to the Akt E17K Mutant.
Biotin
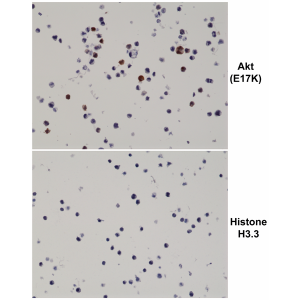
Recombinant Antibody. This antibody reacts to the Akt E17K mutant. No cross reactivity with wild type Akt. Applications: ELISA, IHC, WB. Clone: RM336. Isotype: Rabbit IgG. Formulation: Liquid. 50% Glycerol/PBS with 1% BSA and 0.09% sodium azide. Akt, also referred to as PKB or Rac, plays a critical role in controlling survival and apoptosis. This protein kinase is activated at 2 phosphorylation sites Thr308 and Ser473. Akt promotes cell survival by inhibiting apoptosis through phosphorylation and inactivation of several targets, including Bad, forkhead transcription factors, c-Raf and caspase-9. In addition to its role in survival and glycogen synthesis, Akt is involved in cell cycle regulation. Akt also plays a critical role in cell growth by directly phosphorylating mTOR in a rapamycin-sensitive complex containing raptor. Mutation of the glutamic acid at residue 17 to lysine (E17K) of Akt was initially identified in human breast, colorectal and ovarian cancers. This conserved glutamic acid residue is located at the lipid-binding pocket of the Akt plextrin homology domain. The E17K mutation increases the affinity between Akt and phospholipids at the plasma membrane, leading to increased Akt recruitment, super-activation of the Akt pathway, cellular transformation and tumor formation. Additional studies detect the presence of the Akt (E17K) mutation in multiple cancers, including lung cancer, prostate cancer, endometrial carcinoma and several melanomas.
P31749
Vial
Akt, also referred to as PKB or Rac, plays a critical role in controlling survival and apoptosis. This protein kinase is activated at 2 phosphorylation sites Thr308 and Ser473. Akt promotes cell survival by inhibiting apoptosis through phosphorylation and inactivation of several targets, including Bad, forkhead transcription factors, c-Raf and caspase-9. In addition to its role in survival and glycogen synthesis, Akt is involved in cell cycle regulation. Akt also plays a critical role in cell growth by directly phosphorylating mTOR in a rapamycin-sensitive complex containing raptor. Mutation of the glutamic acid at residue 17 to lysine (E17K) of Akt was initially identified in human breast, colorectal and ovarian cancers. This conserved glutamic acid residue is located at the lipid-binding pocket of the Akt plextrin homology domain. The E17K mutation increases the affinity between Akt and phospholipids at the plasma membrane, leading to increased Akt recruitment, super-activation of the Akt pathway, cellular transformation and tumor formation. Additional studies detect the presence of the Akt (E17K) mutation in multiple cancers, including lung cancer, prostate cancer, endometrial carcinoma and several melanomas.
Protein A purified.
Rabbit
This antibody reacts to the Akt E17K mutant. No cross reactivity with wild type Akt.
Non-hazardous
Primary Antibodies
12352203
Stable for at least 1 year after receipt when stored at -20°C.