Further Information
Alternate Names/Synonyms:
Antibiotic X464; Azalomycin M; Polyetherin A; Antibiotic K178; Helexin C
White to yellow powder.
28643-80-3
32160000
solid
GHS06
Protect from light and moisture.
H301 - H319
InChi=1S/C40H68O11.Na/c1-21-11-12-28(46-33(21)26(6)36(42)43)17-29-18-30(45-10)27(7)40(48-29)25(5)19-38(9,51-40)32-13-14-37(8,49-32)35-23(3)16-31(47-35)34-22(2)15-24(4)39(44,20-41)50-34;/h21-35,41,44H,11-20H2,1-10H3,(H,42,43);/q;+1/p-1/t21-,22-,23-,24+,25+,26+,27+,28+,29+,30+,31+,32+,33+,34-,35+,37-,38-,39-,40+;/m0./s1
MOYOTUKECQMGHE-PDEFJWSRSA-M
Chemical. CAS: 28643-80-3. Formula: C40H67NaO11. MW: 746.94. Nigericin is an antibiotic, used as a high affinity ionophore for monovalent cations such as H+, K+, Na+, Pb2+. It shows antibacterial (Gram-positive), antifungal, antitumor and antiviral activity and disrupts membrane potential of mitochondria. It sprimary use is as a NLRP3 activator in inflammasome research.
MFCD30377206
C40H67NaO11
746.94
Vial
III
P264 - P270 - P280 - P301 + P310 - P305 + P351 + P338 - P337 + P313
Nigericin is an antibiotic, used as a high affinity ionophore for monovalent cations such as H+, K+, Na+, Pb2+. It shows antibacterial (Gram-positive), antifungal, antitumor and antiviral activity and disrupts membrane potential of mitochondria. It sprimary use is as a NLRP3 activator in inflammasome research.
>98% (TLC)
Danger
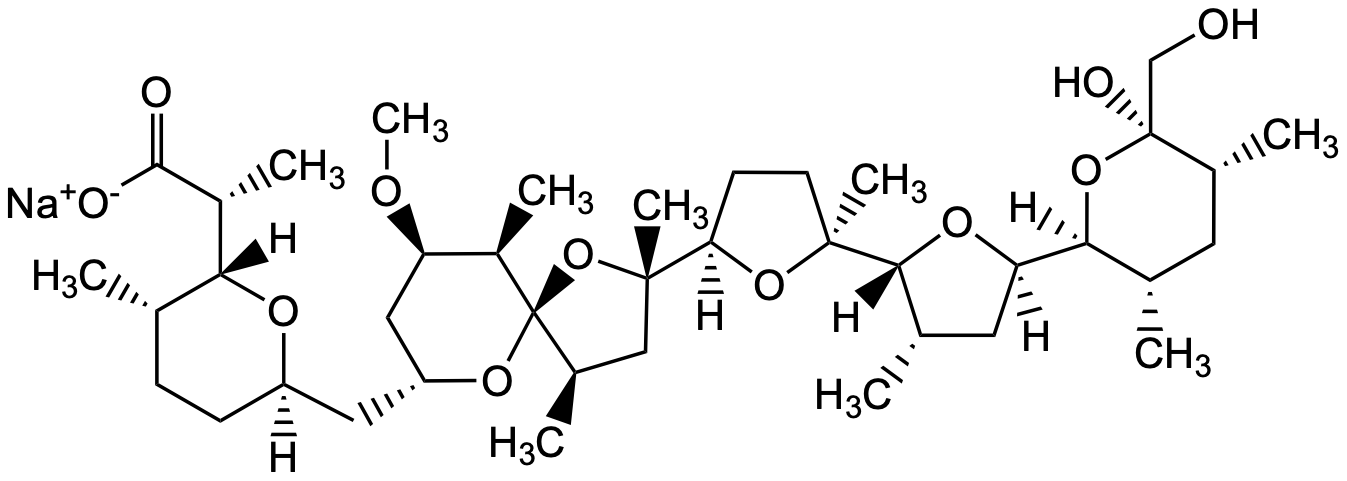
[Na+].[H][C@@]1(CC[C@H](C)[C@@]([H])(O1)[C@@H](C)C([O-])=O)C[C@]2([H])C[C@@H](OC)[C@@H](C)[C@]3(O2)O[C@@](C)(C[C@H]3C)[C@@]4([H])CC[C@](C)(O4)[C@]5([H])O[C@]([H])(C[C@@H]5C)[C@@]6([H])O[C@@](O)(CO)[C@H](C)C[C@@H]6C
Soluble in methanol or ethanol. Insoluble in water.
Synthetic
Excepted Quantity
UN3462
12352200
Stable for at least 2 years after receipt when stored at +4°C.