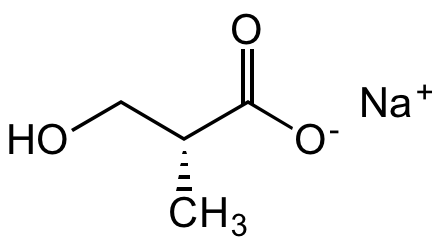
R-3-Hydroxyisobutyric acid sodium salt
| Code | Size | Price |
|---|
| CDX-H0291-M100 | 100 mg | £157.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
Ambient
Storage:
+20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
(R)-beta-Hydroxyisobutyric acid sodium salt; (R)-3-Hydroxy-2-methylpropionic acid sodium salt; R-HIBA; R-beta-HIBA-Na, Sodium (R)-3-hydroxy-2-methylpropionate
Appearance:
White to off-white powder.
CAS:
1228078-57-6
EClass:
32160000
Form (Short):
solid
Handling Advice:
Protect from light and moisture.
InChi:
InChI=1S/C4H8O3.Na/c1-3(2-5)4(6)7;/h3,5H,2H2,1H3,(H,6,7);/q;+1/p-1/t3-;/m1./s1
InChiKey:
RBJZIQZDAZLXEK-AENDTGMFSA-M
Long Description:
Chemical. CAS: 1228078-57-6. Formula: C4H7O3 . Na. MW: 103.1 . 23.0. Synthetic. Intermediate formed in the valine catabolism. It is a reactant of the enzymes involved in branched chain amino acid metabolism 3-hydroxyisobutyrate dehydrogenase and 3-hydroxyisobutyryl-CoA hydrolase and it was found to have increased concentrations in MS patient metabolic profiles. This compound may be used to study the distribution, characterization and kinetics of these enzymes. Shown to inhibit key enzymes of the energy metabolism.
MDL:
MFCD17215934
Molecular Formula:
C4H7O3 . Na
Molecular Weight:
103.1 . 23.0
Package Type:
Vial
Product Description:
Intermediate formed in the valine catabolism. It is a reactant of the enzymes involved in branched chain amino acid metabolism 3-hydroxyisobutyrate dehydrogenase and 3-hydroxyisobutyryl-CoA hydrolase and it was found to have increased concentrations in MS patient metabolic profiles. This compound may be used to study the distribution, characterization and kinetics of these enzymes. Shown to inhibit key enzymes of the energy metabolism.
Purity:
>96% (GC)
SMILES:
C[C@H](CO)C([O-])=O.[Na+]
Solubility Chemicals:
Soluble in water.
Source / Host:
Synthetic
Transportation:
Non-hazardous
UNSPSC Category:
Biochemical Reagents
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at +4°C.
References
(1) S. Landaas; Clin. Chim. Acta 64, 143 (1975) | (2) F. Podebrad, et al.; Clin. Chim. Acta 292, 93 (2000) | (3) J.W. Hawes, et al.; Methods Enzymol. 324, 218 (2000) | (4) F.J. Loupatty, et al.; Am. J. Hum. Genet. 80, 195 (2007) | (5) C.M. Viegas, et al.; Int. J. Dev. Neurosci. 26, 293 (2008) | (6) R.J. Wanders, et al.; J. Inherit. Metab. Dis. 35, 5 (2012)