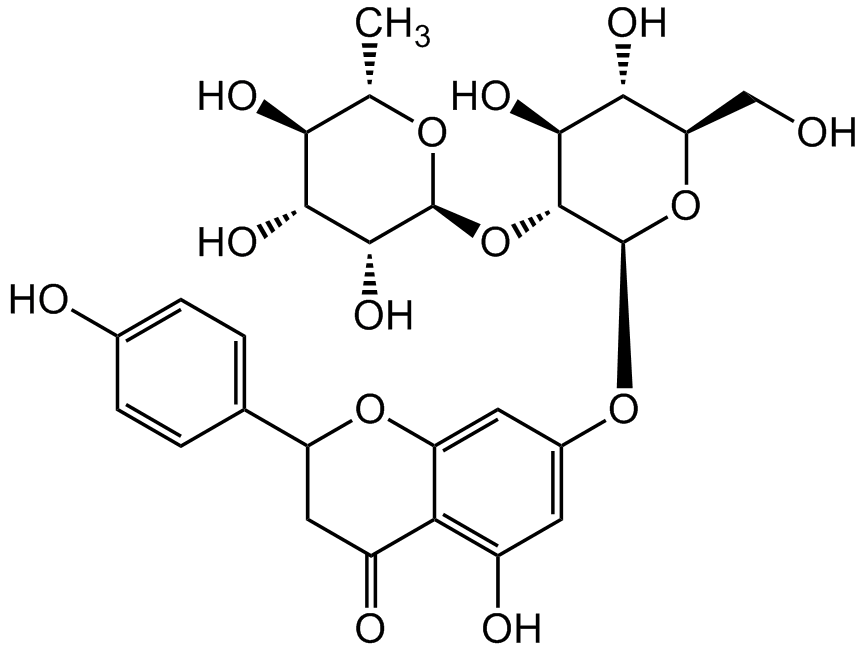
Naringin
Product Code:
CDX-N0234
CDX-N0234
Host Type:
Plant
Plant
Regulatory Status:
RUO
RUO
Shipping:
AMBIENT
AMBIENT
Storage:
Short Term: +20°C Long Term: +4°C
Short Term: +20°C Long Term: +4°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-N0234-G100 | 100 g | £108.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Alternate Names/Synonyms:
4',5,7-Trihydroxyflavanone 7-rhamnoglucoside; Naringenin 7-neohesperidoside; Naringenine-7-rhamnosidoglucoside; Naringoside; NSC 5548
Appearance:
Light yellow to beige powder.
CAS:
10236-47-2
EClass:
32160000
Form (Short):
solid
GHS Symbol:
GHS07
Handling Advice:
Protect from light and moisture.
Hazards:
H315-H319-H335
InChi:
InChI=1S/C27H32O14/c1-10-20(32)22(34)24(36)26(37-10)41-25-23(35)21(33)18(9-28)40-27(25)38-13-6-14(30)19-15(31)8-16(39-17(19)7-13)11-2-4-12(29)5-3-11/h2-7,10,16,18,20-30,32-36H,8-9H2,1H3/t10-,16?,18+,20-,21+,22+,23-,24+,25+,26-,27+/m0/s1
InChiKey:
DFPMSGMNTNDNHN-JJLSSNRUSA-N
Long Description:
Chemical. CAS: 10236-47-2. Formula: C27H32O14. MW: 580.54. Naringin is a citrus-derived flavonoid (bitter principal). It undergoes extensive metabolism in the liver. It has a broad panel of properties, including anti-inflammatory, antioxidant, anticancer, antidiabetic, cardioprotective, bone regenerative and neuroprotective activities. Naringin acts by modulating several signaling pathway targets, activity is primarily attributed to its anti-inflammatory (via inhibiting recruitment of cytokines and inflammatory transcription factors) and antioxidant (via scavenging of free radicals, bolstering of endogenous antioxidant defense system and metal ion chelation) effects. It increases the expression of neurotrophic factor in dopaminergic neurons, providing neuroprotection.
MDL:
MFCD00148888
Molecular Formula:
C27H32O14
Molecular Weight:
580.54
Package Type:
Vial
Precautions:
P261-P305 + P351 + P338
Product Description:
Naringin is a citrus-derived flavonoid (bitter principal). It undergoes extensive metabolism in the liver. It has a broad panel of properties, including anti-inflammatory, antioxidant, anticancer, antidiabetic, cardioprotective, bone regenerative and neuroprotective activities. Naringin acts by modulating several signaling pathway targets, activity is primarily attributed to its anti-inflammatory (via inhibiting recruitment of cytokines and inflammatory transcription factors) and antioxidant (via scavenging of free radicals, bolstering of endogenous antioxidant defense system and metal ion chelation) effects. It increases the expression of neurotrophic factor in dopaminergic neurons, providing neuroprotection.
Purity:
>98% (HPLC)
Signal Word:
Warning
SMILES:
OC1=C2C(OC(C3=CC=C(O)C=C3)CC2=O)=CC(O[C@@H]4O[C@H](CO)[C@@H](O)[C@H](O)[C@H]4O[C@@H]5O[C@@H](C)[C@H](O)[C@@H](O)[C@H]5O)=C1
Solubility Chemicals:
Soluble in DMSO (10mg/ml), DMF (20mg/ml) or ethanol (1mg/ml).
Source / Host:
Plant
Transportation:
Non-hazardous
UNSPSC Category:
Natural Products/Extracts
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at +4°C.
Documents
References
(1) U. Fuhr, et al.; Br. J. Clin. Pharmacol. 35, 431 (1993) | (2) P.C. Ho & D.J. Saville; J. Pharm. Pharmaceut. Sci. 4, 217 (2001) | (3) S. Bharti, et al.; Planta Med. 80, 437 (2014) (Review) | (4) U.J. Jung, et al.; Exp. Neurobiol. 23, 124 (2014) (Review) | (5) M.A. Alam, et al.; Adv. Nutr. 5, 404 (2014) (Review) | (6) R. Chen, et al.; Pharm. Biol. 54, 3203 (2016) (Review) | Anti-estrogenic and anti-aromatase activities of citrus peels major compounds in breast cancer: D.M. El-Kersh, et al.; Nature Sci. Rep. 11, 7121 (2021)