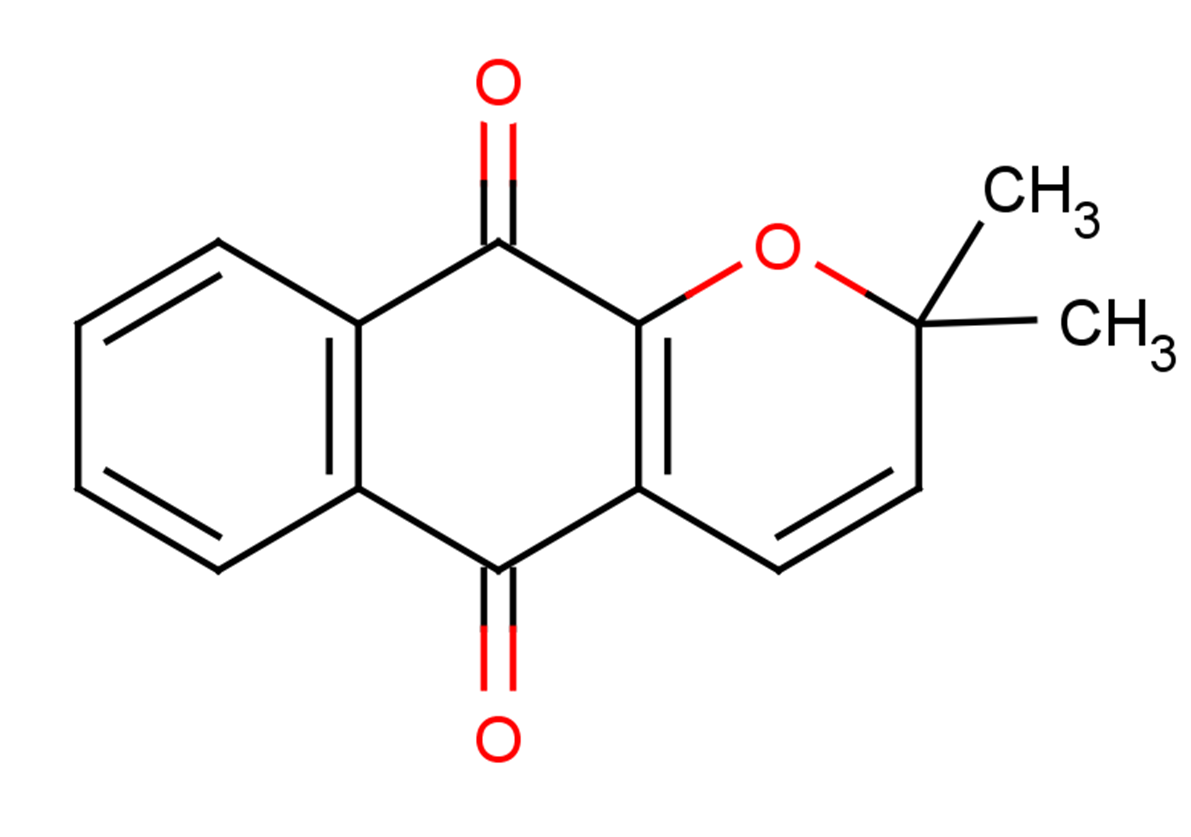
Xyloidone
| Code | Size | Price |
|---|
| TAR-T19962-50mg | 50mg | Enquire | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T19962-1mg | 1mg | £164.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T19962-5mg | 5mg | £287.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T19962-10mg | 10mg | £388.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T19962-25mg | 25mg | £600.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Xyloidone exhibits antifungal activity and completely inhibits the mycelial growth of B. cinerea, Colletotrichum acutatum Simmonds, Colletotrichum gloeosporioides Simmonds, M. grisea, and Pythium ultimum Trow over a range of 0.4-33.3 mg/L.
CAS:
15297-92-4
Formula:
C15H12O3
Molecular Weight:
240.258
Pathway:
Microbiology/Virology
Purity:
0.9861
SMILES:
CC1(C)OC2=C(C=C1)C(=O)c1ccccc1C2=O
Target:
Antifungal
References
1. Moraes DC, et al. ?-lapachone and ?-nor-lapachone modulate Candida albicans viability and virulence factors. J Mycol Med. 2018 Mar 26. pii: S1156-5233(17)30379-7.
2. Garkavtsev I, et al. Dehydro-alpha-lapachone, a plant product with antivascular activity. Proc Natl Acad Sci U S A. 2011 Jul 12;108(28):11596-601.
3. Cho JY, et al. Dehydro-alpha-lapachone isolated from Catalpa ovata stems: activity against plant pathogenic fungi. Pest Manag Sci. 2006 May;62(5):414-8.
4. Otten S, Rosazza JP. Microbial transformations of natural antitumor agents: conversion of lapachol to dehydro-alpha-lapachone by Curvularia lunata. Appl Environ Microbiol. 1979 Aug;38(2):311-3.