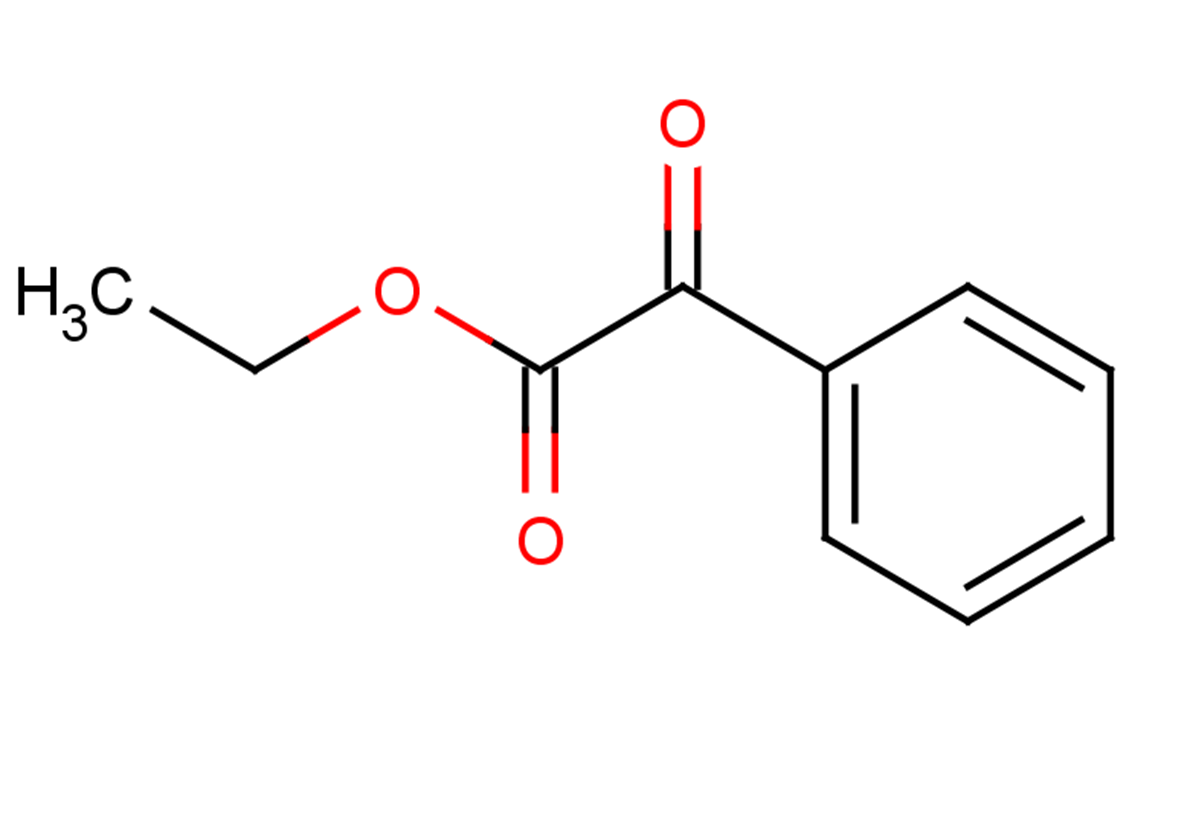
Ethyl phenylglyoxylate
| Code | Size | Price |
|---|
| TAR-T20351-1g | 1g | £94.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Ethyl phenylglyoxylate is a simultaneous inhibitor and substrate of chicken liver carboxylesterase.
CAS:
1603-79-8
Formula:
C10H10O3
Molecular Weight:
178.187
Pathway:
Metabolism
Purity:
0.9897
SMILES:
CCOC(=O)C(=O)c1ccccc1
Target:
Lipid
References
1. Sztanke K. Synthesis of new derivatives of 8-aryl-3-phenyl-6,7-dihydro-4H-imidazo[2, 1-c][1,2,4]triazin-4-one. Acta Pol Pharm. 2004 Sep-Oct;61(5):373-7.
2. Hu S, Neckers DC. Photochemical Reactions of Alkyl Phenylglyoxylates(1). J Org Chem. 1996 Sep 6;61(18):6407-6415.
3. Bentabed-Ababsa G, et al. Polar [3 + 2] cycloaddition of ketones with electrophilically activated carbonyl ylides. Synthesis of spirocyclic dioxolane indolinones. Org Biomol Chem. 2008 Sep 7;6(17):3144-57.
4. Tokuda O, et al. A practical synthesis of (S)-2-cyclohexyl-2-phenylglycolic acid via organocatalytic asymmetric construction of a tetrasubstituted carbon center. Org Lett. 2005 Oct 27;7(22):5103-5.