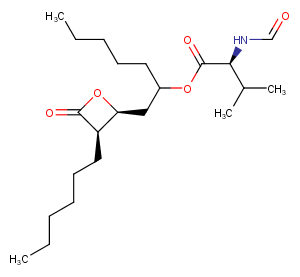
Valilactone
| Code | Size | Price |
|---|
| TAR-T29091-5mg | 5mg | £850.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Valilactone is a fatty acid synthase inhibitor, which is produced by a cultured strain of soil actinomycetes.
CAS:
113276-96-3
Formula:
C22H39NO5
Molecular Weight:
397.556
Purity:
0.98
SMILES:
CCCCCC[C@@H]1[C@H](CC(CCCCC)OC(=O)[C@@H](NC=O)C(C)C)OC1=O
References
1. Richardson RD, Ma G, Oyola Y, Zancanella M, Knowles LM, Cieplak P, Romo D, Smith JW. Synthesis of novel beta-lactone inhibitors of fatty acid synthase. J Med Chem. 2008 Sep 11;51(17):5285-96. doi: 10.1021/jm800321h. PubMed PMID: 18710210; PubMed Central PMCID: PMC3172131.
2. Ma G, Zancanella M, Oyola Y, Richardson RD, Smith JW, Romo D. Total synthesis and comparative analysis of orlistat, valilactone, and a transposed orlistat derivative: Inhibitors of fatty acid synthase. Org Lett. 2006 Sep 28;8(20):4497-500. PubMed PMID: 16986934.
3. Wu Y, Sun YP. An expeditious enantioselective total synthesis of valilactone. J Org Chem. 2006 Jul 21;71(15):5748-51. PubMed PMID: 16839158.
4. Yoshinari K, Aoki M, Ohtsuka T, Nakayama N, Itezono Y, Mutoh M, Watanabe J, Yokose K. Panclicins, novel pancreatic lipase inhibitors. II. Structural elucidation. J Antibiot (Tokyo). 1994 Dec;47(12):1376-84. PubMed PMID: 7844032.