CX-659S
| Code | Size | Price |
|---|
| TAR-T31126-5mg | 5mg | £850.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T31126-50mg | 50mg | £1,661.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T31126-100mg | 100mg | £2,079.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
CX-659S is a novel diaminuracil derivative that indirectly inhibits Langerhans cell function by blocking the MEK1/2 -- ERK1/2 pathway in keratinocytes. CX-659S can inhibit the contact hypersensitivity induced by haptens in mice.
CAS:
204980-81-4
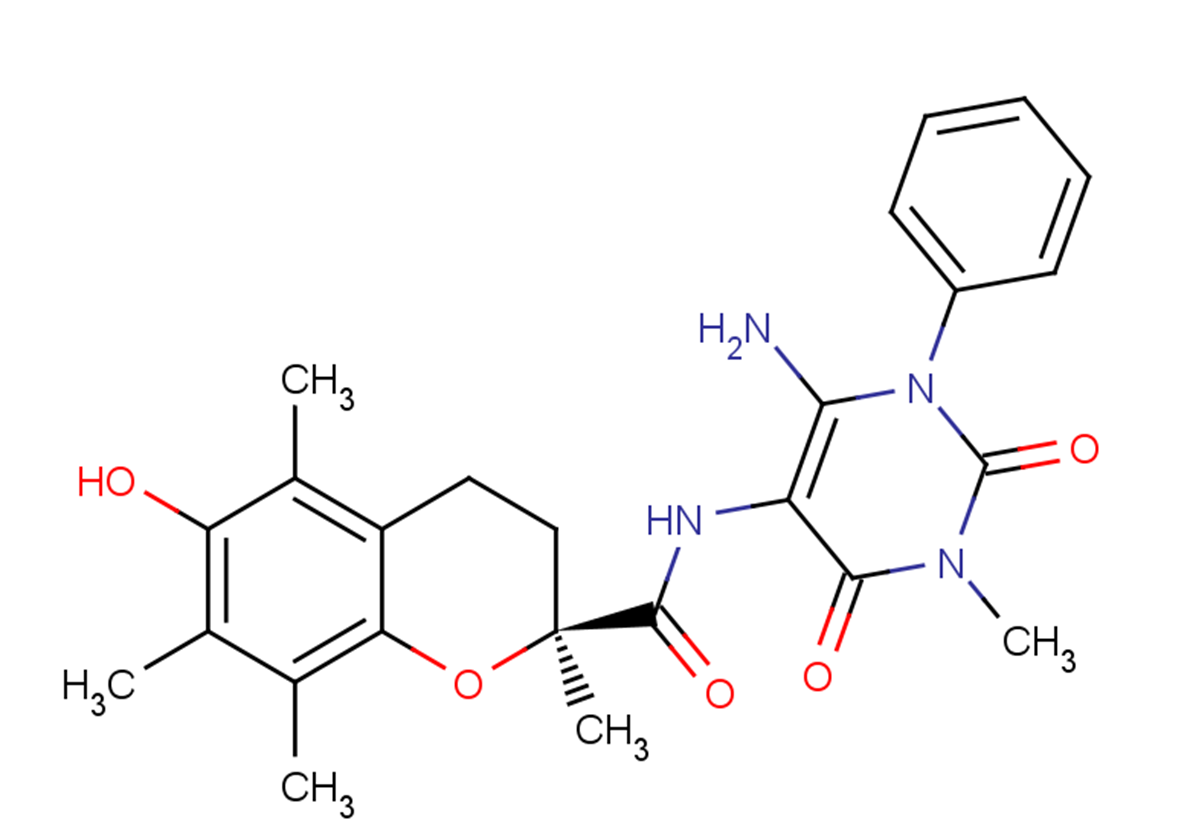
Formula:
C25H28N4O5
Molecular Weight:
464.522
Purity:
0.98
SMILES:
Cc1c(C)c2O[C@@](C)(CCc2c(C)c1O)C(=O)Nc1c(N)n(-c2ccccc2)c(=O)n(C)c1=O
References
1. Tobe M, Tsuboi K, Hasegawa F, Fujiwara N, Inoue Y, Isobe M, Isobe Y. Synthesis and biological evaluation of novel orally available 1-phenyl-6-aminouracils containing dimethyldihydrobenzofuranol structure for the treatment of allergic skin diseases. Bioorg Med Chem Lett. 2016 Feb 15;26(4):1292-5. doi: 10.1016/j.bmcl.2016.01.019. Epub 2016 Jan 8. PubMed PMID: 26786694.
2. Kido M, Takeuchi S, Esaki H, Hayashida S, Furue M. Scratching behavior does not necessarily correlate with epidermal nerve fiber sprouting or inflammatory cell infiltration. J Dermatol Sci. 2010 May;58(2):130-5. doi: 10.1016/j.jdermsci.2010.03.007. Epub 2010 Mar 19. PubMed PMID: 20399618.
3. Ohmori K, Adachi K, Manabe H, Harada D, Ohshima E. [Recent development of new drugs for the treatment of allergic diseases]. Nihon Yakurigaku Zasshi. 2004 May;123(5):335-48. Review. Japanese. PubMed PMID: 15118258.
4. Isobe Y, Tobe M, Inoue Y, Isobe M, Tsuchiya M, Hayashi H. Structure and activity relationships of novel uracil derivatives as topical anti-inflammatory agents. Bioorg Med Chem. 2003 Nov 17;11(23):4933-40. PubMed PMID: 14604654.