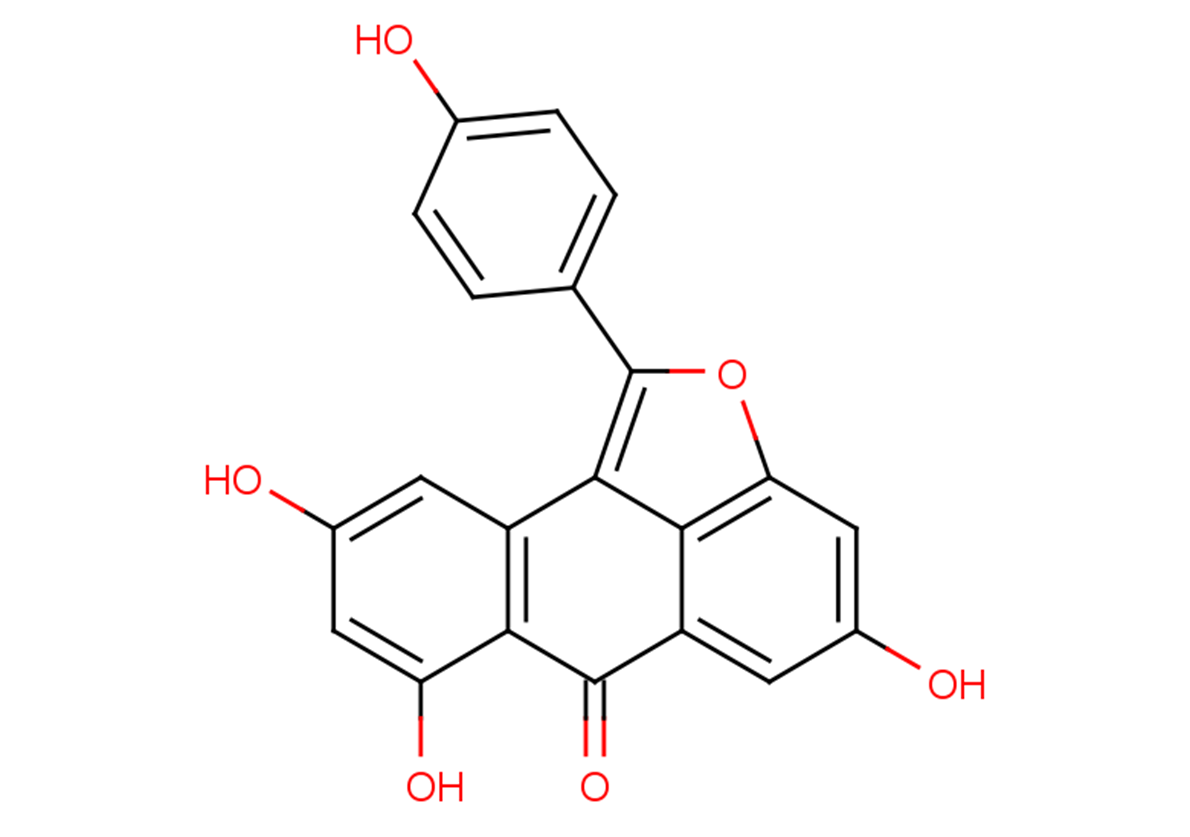
Diptoindonesin G
| Code | Size | Price |
|---|
| TAR-T31523-5mg | 5mg | £850.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T31523-50mg | 50mg | £1,661.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T31523-100mg | 100mg | £2,079.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Diptoindonesin G is an ER? stabilizer that regulates the stability of ERα and ER? by targeting CHIP E3 ubiquitin ligase. Diptoindonesin G could significantly increase the protein stability of ER?, and reduce the protein level of ERα,
CAS:
1190948-58-3
Formula:
C21H12O6
Molecular Weight:
360.321
Purity:
0.98
SMILES:
Oc1ccc(cc1)-c1oc2cc(O)cc3C(=O)c4c(O)cc(O)cc4-c1c23
References
1. Xu T, Sun T, Zhang Y, Qiu B, Wang Y, Qin Y, Dong G. RhI-Catalyzed Carboacylation/Aromatization Cascade Initiated by Regioselective C-C Activation of Benzocyclobutenones. Angew Chem Int Ed Engl. 2018 Jan 23. doi: 10.1002/anie.201713179. [Epub ahead of print] PubMed PMID: 29360217.
2. Singh DK, Kim I. Skeletal Reorganization: Synthesis of Diptoindonesin G from Pauciflorol F. J Org Chem. 2018 Jan 24. doi: 10.1021/acs.joc.7b03089. [Epub ahead of print] PubMed PMID: 29322777.
3. Gao J, Fan M, Xiang G, Wang J, Zhang X, Guo W, Wu X, Sun Y, Gu Y, Ge H, Tan R, Qiu H, Shen Y, Xu Q. Diptoindonesin G promotes ERK-mediated nuclear translocation of p-STAT1 (Ser727) and cell differentiation in AML cells. Cell Death Dis. 2017 May 4;8(5):e2765. doi: 10.1038/cddis.2017.159. PubMed PMID: 28471454; PubMed Central PMCID: PMC5520695.
4. Jung Y, Singh DK, Kim I. Symmetry-based approach to oligostilbenoids: Rapid entry to viniferifuran, shoreaphenol, malibatol A, and diptoindonesin G. Beilstein J Org Chem. 2016 Dec 12;12:2689-2693. doi: 10.3762/bjoc.12.266. eCollection 2016. PubMed PMID: 28144340; PubMed Central PMCID: PMC5238531.