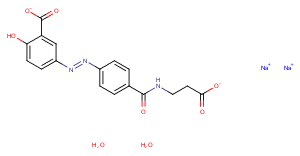
Balsalazide sodium hydrate
| Code | Size | Price |
|---|
| TAR-T4615-25mg | 25mg | £176.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T4615-100mg | 100mg | £315.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Balsalazide is an anti-inflammatory compound used in the treatment of Inflammatory Bowel Disease.Balsalazide is a new 5-aminosalicylic acid (5-ASA) containing prodrug. High dose balsalazide (3.0 g twice daily) was superior in maintaining remission in patients with ulcerative colitis compared with a low dose (1.5 g twice daily) or a standard dose of mesalazine (0.5 g three times daily). All three treatments were safe and well tolerated [1]. Balsalazide is approved for the treatment of mild-to-moderate active UC. It is efficacious for the induction of remission in mild to moderate UC and has a favorable safety profile, with the added advantages of greater efficacy of remission induction and rapidity of onset [2].
CAS:
150399-21-6
Formula:
C17H17N3Na2O8
Molecular Weight:
437.316
Pathway:
Immunology/Inflammation; Stem Cells; JAK/STAT signaling
Purity:
0.9967
SMILES:
O.O.[Na+].[Na+].Oc1ccc(cc1C([O-])=O)N=Nc1ccc(cc1)C(=O)NCCC([O-])=O
Target:
STAT; Interleukin
References
Wiggins JB, et al. Balsalazide: a novel 5-aminosalicylate prodrug for the treatment of active ulcerative colitis. Expert Opin Drug Metab Toxicol, 2009. 5(10): p. 1279-1284.
Kruis W, et al. Low dose balsalazide (1.5 g twice daily) and mesalazine (0.5 g three times daily) maintained remission of ulcerative colitis but high dose balsalazide (3.0 g twice daily) was superior in preventing relapses. Gut, 2001. 49(6): p. 783-789.