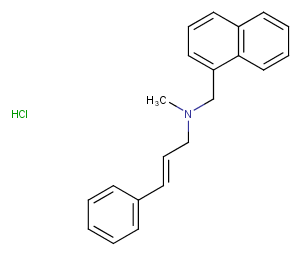
Naftifine hydrochloride
| Code | Size | Price |
|---|
| TAR-T1543-50mg | 50mg | £97.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T1543-100mg | 100mg | £109.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T1543-500mg | 500mg | £145.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T1543-1g | 1g | £179.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Naftifine Hydrochloride is the hydrochloride salt form of naftifine, an allylamine derivate with synthetic broad-spectrum antifungal activity. Although the exact mechanism through which naftifine hydrochloride exerts its effect is unknown, it appears to selectively inhibit the enzyme squalene 2, 3-epoxidase, thereby inhibiting the biosynthesis of sterol. This results in a decreased amount of sterols, especially ergosterol which is the primary fungal membrane sterol, and a corresponding accumulation of squalene in fungal cells. Naftifine hydrochloride can be fungicidal as well as fungistatic to yeasts depending on the concentration and the organisms involved.
CAS:
65473-14-5
Formula:
C21H22ClN
Molecular Weight:
323.86
Pathway:
Microbiology/Virology; GPCR/G Protein; Stem Cells
Purity:
0.9984
SMILES:
Cl.CN(CC=Cc1ccccc1)Cc1cccc2ccccc12
Target:
Hedgehog/Smoothened; Antibiotic; Antifungal
References
1. Georgopoulos A, et al. Antimicrob Agents Chemother, 1981, 19(3), 386-389.
2. Ryder NS, et al. Antimicrob Agents Chemother, 1984, 25(4), 483-487.
3. Parish LC, et al. J Drugs Dermatol, 2011, 10(10), 1142-1147.
4. Gupta AK, et al. J Cutan Med Surg, 2008, 12(2), 51-58.