Anti-Human PHAP I (CT)
| Code | Size | Price |
|---|
| LEI-P209-20ug | 20 ug | £199.00 |
Quantity:
| LEI-P209-0.1mg | 0.1 mg | £591.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Host Type: Rabbit
Antibody Clonality: Polyclonal
Regulatory Status: RUO
Target Species: Human
Applications:
- Immunohistochemistry- Paraffin Embedded (IHC-P)
- Western Blot (WB)
Shipping:
Ambient
Storage:
This polyclonal antibody is stable for at least one week when stored at 2-8°C. For long term storage aliquot in working volumes without diluting and store at -20°C in a manual defrost freezer. Avoid Repeated Freeze Thaw Cycles.
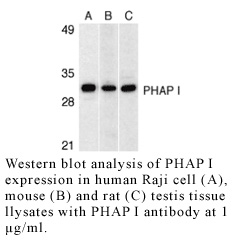
Images
Further Information
Concentration:
0.5 mg/ml
Conjugate/Tag/Label:
Purified No Carrier Protein
Format:
This polyclonal antibody is formulated in phosphate buffered saline (PBS) pH 7.4 containing 0.02% sodium azide as a preservative.
Formulation:
This polyclonal antibody is formulated in phosphate buffered saline (PBS) pH 7.4 containing 0.02% sodium azide as a preservative.
Immunogen:
PN:P219
Target:
PHAP I
References
1. Jiang, X. et al. (2003) Science 299(5604):223 2. Nicholson, DW. et al. (2003) Science 299(5604):214
Related Products
| Product Name | Product Code | Supplier | Anti-Human PHAP I (Intermediate Domain) | LEI-P210 | Leinco Technologies | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PHAP I (CT) Blocking Peptide | LEI-P219 | Leinco Technologies | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHAP I (Intermediate Domain) Blocking Peptide | LEI-P220 | Leinco Technologies | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||