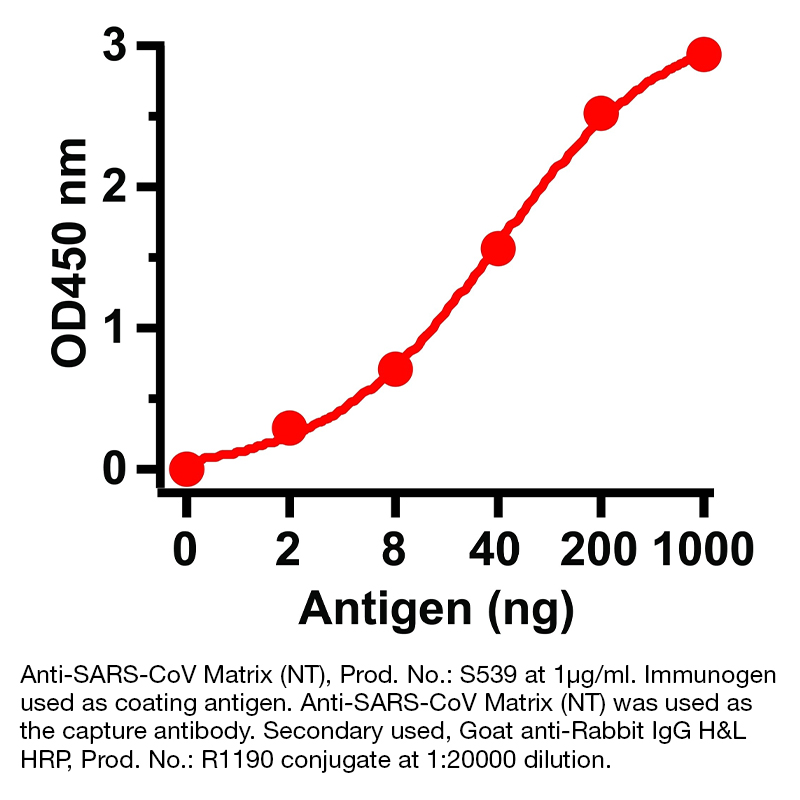
Anti-SARS-CoV Matrix (NT)
| Code | Size | Price |
|---|
| LEI-S539-20ug | 20 ug | £199.00 |
Quantity:
| LEI-S539-0.1mg | 0.1 mg | £591.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Host Type: Rabbit
Antibody Isotype: IgG
Antibody Clonality: Polyclonal
Regulatory Status: RUO
Target Species: Virus
Application: Enzyme-Linked Immunosorbent Assay (ELISA)
Shipping:
Ambient
Storage:
This polyclonal antibody is stable for at least one week when stored at 2-8°C. For long term storage aliquot in working volumes without diluting and store at -20°C in a manual defrost freezer. Avoid Repeated Freeze Thaw Cycles.
Images
Further Information
Concentration:
0.5 mg/ml
Conjugate/Tag/Label:
Purified No Carrier Protein
Format:
This polyclonal antibody is formulated in phosphate buffered saline (PBS) pH 7.4 containing 0.02% sodium azide as a preservative.
Formulation:
This polyclonal antibody is formulated in phosphate buffered saline (PBS) pH 7.4 containing 0.02% sodium azide as a preservative.
Immunogen:
Anti-SARS-CoV Matrix antibody was raised against a peptide corresponding to 13 amino acids near the amino-terminus of SARS-CoV Matrix protein. The immunogen is located within the first 50 amino acids of SARS-CoV Matrix.
Long Description:
A coronavirus, SARS (Severe Acute Respiratory Syndrome), was first identified 2002-2003. Coronaviruses are a major cause of upper respiratory diseases in humans. The genomes of these viruses are positive-stranded RNA approximately 27-31kb in length. The M protein (Membrane protein, Matrix protein) is one of the major structural viral proteins. It is an integral membrane protein involved in the budding of the viral particles and interacts with S (Spike) protein and the nucleocapsid protein.
Target:
SARS M
References
1. Marra, M. A. et al. (2003) Science 300:1399
2. Rota, P. A. et al. (2003) Science 300:1394
3. Navas-Nartin, S. R. et al. (2004) J. Neurovirol. 10:75
4. Opstelten, D. J. et al. (1995) J. Cell Biol. 131:339
Related Products
| Product Name | Product Code | Supplier | Anti-SARS-CoV Matrix (CT) | LEI-S538 | Leinco Technologies | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Severe Acute Respiratory Syndrome M (CT) Blocking Peptide | LEI-S547 | Leinco Technologies | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Severe Acute Respiratory Syndrome M (NT) Blocking Peptide | LEI-S548 | Leinco Technologies | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||