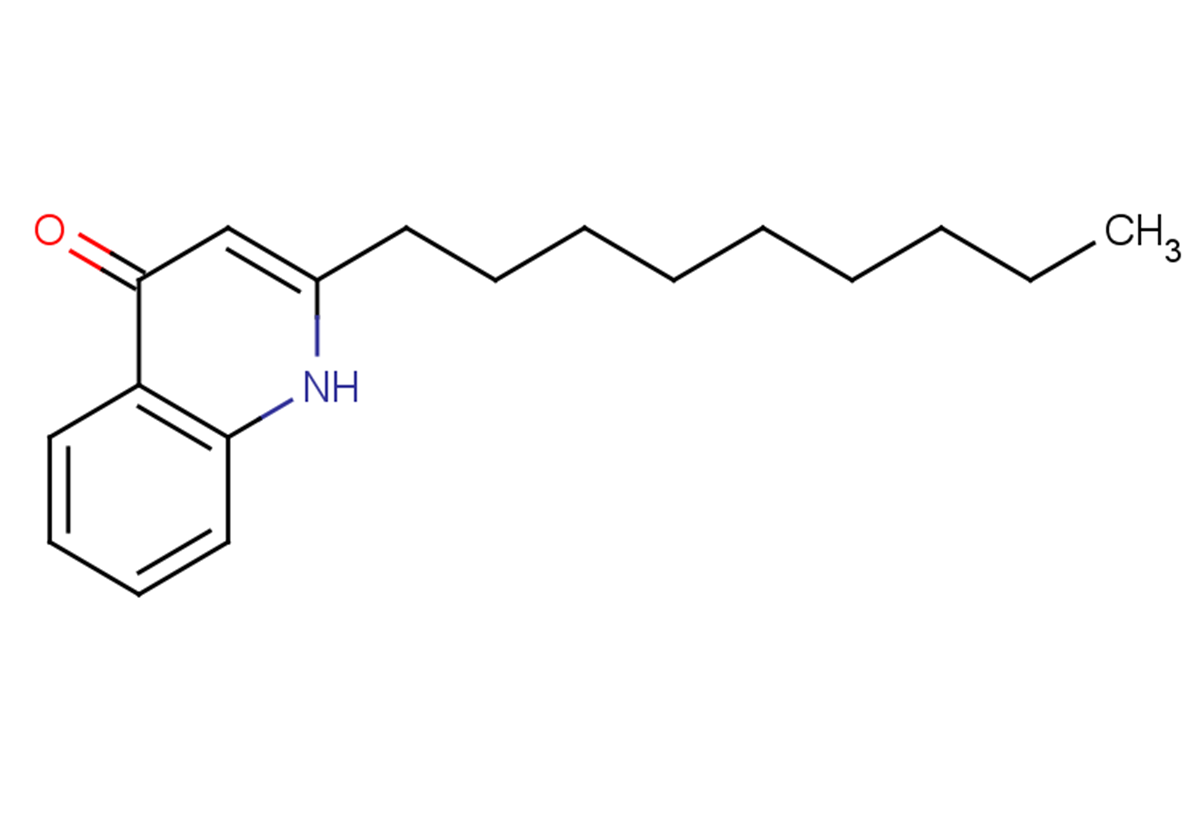
2-Nonylquinolin-4(1H)-one
| Code | Size | Price |
|---|
| TAR-T38375-1mg | 1mg | £227.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T38375-5mg | 5mg | £416.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T38375-10mg | 10mg | £581.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T38375-25mg | 25mg | £863.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T38375-50mg | 50mg | £1,163.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T38375-100mg | 100mg | £1,540.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20°C
Images
Documents
Further Information
Bioactivity:
2-Nonylquinolin-4(1H)-one reduces infection of Huh 7.5 cells by hepatitis C virus (HCV) with an IC50 of 1.4 ug/ml.
CAS:
55396-45-7
Formula:
C18H25NO
Molecular Weight:
271.4
Pathway:
Microbiology/Virology|Proteases/Proteasome
Purity:
0.98
SMILES:
O=C1C=C(NC=2C=CC=CC12)CCCCCCCCC
Target:
HCV Protease
References
Wahyuni, T.S., Widyawaruyanti, A., Lusida, M.I., et al.Inhibition of hepatitis C virus replication by chalepin and pseudane IX isolated from Ruta angustifolia leaves.Fitoterapia.99276-283(2014)
Biavatti, M.W., Vieira, P.C., da Silva, F.d.G.F., et al.Biological activity of quinoline alkaloids from Raulinoa echinata and x-ray structure of flindersiamine.J. Braz. Chem. Soc.13(1)66-70(2002)
Ortori, C.A., Dubern, J.-F., Chhabra, S.R., et al.Simultaneous quantitative profiling of N-acyl-L-homoserine lactone and 2-alkyl-4(1H)-quinolone families of quorum-sensing signaling molecules using LC-MS/MSAnal. Bioanal. Chem.399(2)839-850(2011)
Jin, H.Z., Lee, J.H., Lee, D., et al.Quinolone alkaloids with inhibitory activity against nuclear factor of activated T cells from the fruits of Evodia rutaecarpaBiol. Pharm. Bull.27(6)926-928(2004)