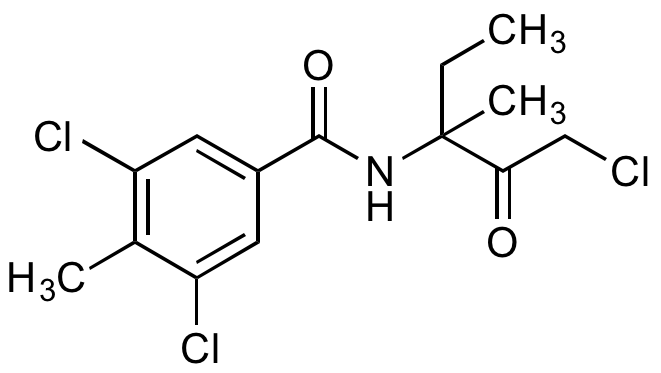
Zoxamide
| Code | Size | Price |
|---|
| CDX-Z0015-M100 | 100 mg | £157.00 |
Quantity:
| CDX-Z0015-M500 | 500 mg | £596.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
Ambient
Storage:
+20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
Zoxium; RH 7281; 3,5-Dichloro-N-(3-chloro-1-ethyl-1-methyl-2-oxopropyl)-4-methylbenzamide
Appearance:
White powder.
CAS:
156052-68-5
EClass:
32160000
Form (Short):
liquid
GHS Symbol:
GHS07,GHS09
Handling Advice:
Keep cool and dry.Protect from light and moisture.
Hazards:
H317, H319, H410
InChi:
InChI=1S/C14H16Cl3NO2/c1-4-14(3,12(19)7-15)18-13(20)9-5-10(16)8(2)11(17)6-9/h5-6H,4,7H2,1-3H3,(H,18,20)
InChiKey:
SOUGWDPPRBKJEX-UHFFFAOYSA-N
Long Description:
Chemical. CAS: 156052-68-5. Formula: C14H16Cl3NO2. MW: 336.6. Synthetic. Fungicide. Primarily an oomycete fungicide although it has some activity against ascomycetes and basidiomycetes. The product acts by disruption of the microtubule structure and inhibition of cell division. The mechanism for this activity is through the covalent binding of the active ingredient to beta-tubulin in the cytoskeleton. Scanning microscopy studies showed zoxamide action against blight on potato crops begins after germination, achieving inhibition of hyphal development and sporangia production. This novel mode of action makes zoxamide an important new tool in antiresistance strategies. Can be used as a reference compound.
MDL:
MFCD03792722
Molecular Formula:
C14H16Cl3NO2
Molecular Weight:
336.6
Package Type:
Vial
Precautions:
P261, P280, P302, P352, P305, P351, P338
Product Description:
Fungicide. Primarily an oomycete fungicide although it has some activity against ascomycetes and basidiomycetes. The product acts by disruption of the microtubule structure and inhibition of cell division. The mechanism for this activity is through the covalent binding of the active ingredient to beta-tubulin in the cytoskeleton. Scanning microscopy studies showed zoxamide action against blight on potato crops begins after germination, achieving inhibition of hyphal development and sporangia production. This novel mode of action makes zoxamide an important new tool in antiresistance strategies. Can be used as a reference compound.
Purity:
>98% (HPLC)
Signal word:
Warning
SMILES:
CCC(C)(NC(=O)C1=CC(Cl)=C(C)C(Cl)=C1)C(=O)CCl
Solubility Chemicals:
Soluble in acetone.
Source / Host:
Synthetic.
Transportation:
Non-hazardous
UNSPSC Category:
Biochemical Reagents
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at +4°C.
References
(1) D.H. Young, et al.; Pest. Manag. Sci. 57, 1081 (2001) | (2) F. Oesch, et al.; Xenobiotica 40, 72 (2010)
Related Products
| Product Name | Product Code | Supplier | Proquinazid | CDX-P0126 | Chemodex | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tetraconazole | CDX-T0115 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thifluzamide | CDX-T0118 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||