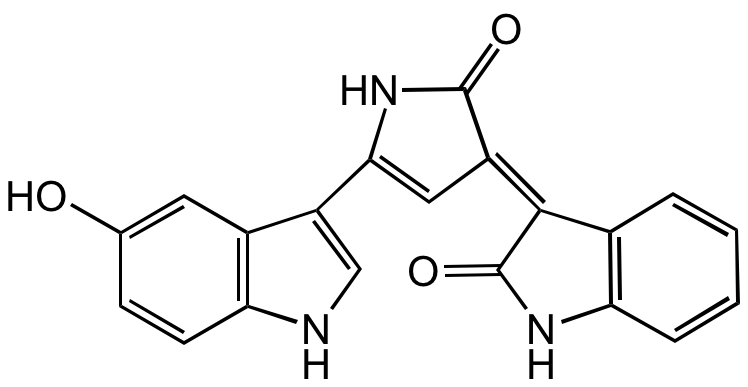
Violacein
| Code | Size | Price |
|---|
| BVT-0473-M001 | 1 mg | £425.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
Ambient
Storage:
Short Term: +4°C Long Term: -20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
Anorosin
Appearance:
Dark purple to black solid.
CAS:
548-54-9
EClass:
32160000
Form (Short):
solid
Handling Advice:
Protect from light.
InChi:
InChI=1S/C20H13N3O3/c24-10-5-6-15-12(7-10)14(9-21-15)17-8-13(19(25)23-17)18-11-3-1-2-4-16(11)22-20(18)26/h1-9,21,24H,(H,22,26)(H,23,25)/b18-13+
InChiKey:
XAPNKXIRQFHCHN-QGOAFFKASA-N
Long Description:
Chemical. CAS: 548-54-9. Formula: C20H13N3O3. MW: 343.3. Isolated from Chromobacterium violaceum. Antibacterial, antifungal and antiprotozoal agent. Quorum sensing metabolite. Antitumor compound. Protective against UV-C irradiation. Possesses immunomodulatory, analgesic and antipyretic activity. Indole-derived purple-colored pigment (free of deoxyviolacein).
MDL:
MFCD01718468
Molecular Formula:
C20H13N3O3
Molecular Weight:
343.3
Package Type:
Plastic Vial
Product Description:
Antibacterial, antifungal and antiprotozoal agent. Quorum sensing metabolite. Antitumor compound. Protective against UV-C irradiation. Possesses immunomodulatory, analgesic and antipyretic activity. Antiulcerogenic and antiviral agent. Indole-derived purple-colored pigment (free of deoxyviolacein).
Purity:
>97% (HPLC, NMR)
SMILES:
OC1=CC2=C(NC=C2C2=CC(C(=O)N2)=C2/C(=O)NC3=C2C=CC=C3)C=C1
Solubility Chemicals:
Soluble in DMSO. Partially soluble in methanol. Insoluble in water and usual organic solvents.
Source / Host:
Isolated from Chromobacterium violaceum.
Transportation:
Non-hazardous
UNSPSC Category:
Natural Products/Extracts
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C.
References
The synthesis of violacein and related compounds: J.A. Ballantine, et al.; J. Chem. Soc. [London] 1960, 2292 (1960) | Evaluation of the antiulcerogenic activity of violacein and its modulation by the inclusion complexation with beta-cyclodextrin: N. Duran, et al.; Can. J. Physiol. Pharmacol. 81, 387 (2003) | Cytotoxicity and potential antiviral evaluation of violacein produced by Chromobacterium violaceum: C.R. Andrighetti-Froehner, et al.; Mem. Inst. Oswaldo Cruz. 98, 843 (2003) | Violacein: properties and biological activities: N. Duran, et al.; Biotechn. Appl. Biochem. 48, 127 (2007) | The protective effects of the violacein pigment against UV-C irradiation in Chromobacterium violaceum: A.N. Abboud & A. Arment; Ohio J. Sci. 111, 28 (2013) | Violacein: properties and production of a versatile bacterial pigment: S.Y. Choi, et al.; Biomed Res. Int. 2015, 1 (2015) | Violacein treatment modulates acute and chronic inflammation through the suppression of cytokine production and induction of regulatory T cells: L. Verinaud, et al.; PLoS One 10, e0125409 (2015) | Violacein induces cell death by triggering mitochondrial membrane hyperpolarization in vitro: A.M. Leal, et al.; Microbiol. 15, 1 (2015) | Antifungal activity of violacein purified from a novel strain of Chromobacterium sp. NIIST (MTCC 5522): A. Sasidharan, et al.; J. Microbiol. [Seoul] 53, 694 (2015) | Violacein, an indole-derived purple-colored natural pigment produced by Janthinobacterium lividum, inhibits the growth of head and neck carcinoma cell lines both in vitro and in vivo: L. Masuelli, et al.; Tumor Biol. 37, 3705 (2016) | Violacein induces apoptosis in human breast cancer cells through up regulation of BAX, p53 and down regulation of MDM2: A.A. Alshatwi, et al.; Exp. Toxicol. Pathol. 68, 89 (2016) | Advances in Chromobacterium violaceum and properties of violacein-Its main secondary metabolite: A review: N. Duran, et al.; Biotechnol. Adv. 34, 1030 (2016) | Violacein antimicrobial activity on Staphylococcus epidermidis and synergistic effect on commercially available antibiotics: H.V. Dodou, et al.; J. Appl. Microbiol. 123, 853 (2017) | Violacein and bacterial predation: promising alternatives for priority multidrug resistant human pathogens: S.Y. Choi, et al.; Future Microbiol. 12, 835 (2017) | Antibacterial mode of action of violacein from Chromobacterium violaceum UTM5 against Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA): C.A. Aruldass, et al.; Environ. Sci. Pollut. Res. 25, 5164 (2017) | Action and function of Chromobacterium violaceum in health and disease: Violacein as a promising metabolite to counteract gastroenterological diseases: G. Justo & N. Duran; Best. Pract. Res. Clin. Gastroenterol. 31, 649 (2017) | Antibacterial mode of action of violacein from Chromobacterium violaceum UTM5 against Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA): C.A. Aruldass, et al.; Environ. Sci. Pollut. R. 25, 5164 (2018) | Prodigiosin, violacein, and volatile organic compounds produced by widespread cutaneous bacteria of amphibians can inhibit two batrachochytrium fungal pathogens: D.C. Woodhams, et al.; Microb. Ecol. 75, 1049 (2018) | Impact of violacein from Chromobacterium violaceum on the mammalian gut microbiome: H. Pauer, et al.; PLoS One 13, e0203748/1 (2018) | Violacein Targets the Cytoplasmic Membrane of Bacteria: A. Cauz, et al.; ACS Infect. Dis. 5, 539 (2019) | Violacein antimicrobial activity on Staphylococcus epidermidis biofilm: H.V. Dodou, et al.; Nat. Prod. Res. Online (2019) | The bacterial product violacein exerts an immunostimulatory effect via TLR8: F. Venegas, et al.; Sci. Rep. 9, 1 (2019) | Antichagasic effect of violacein from Chromobacterium violaceum: J.A. Canuto, et al.; J. Appl. Microbiol. 127, 1373 (2019)