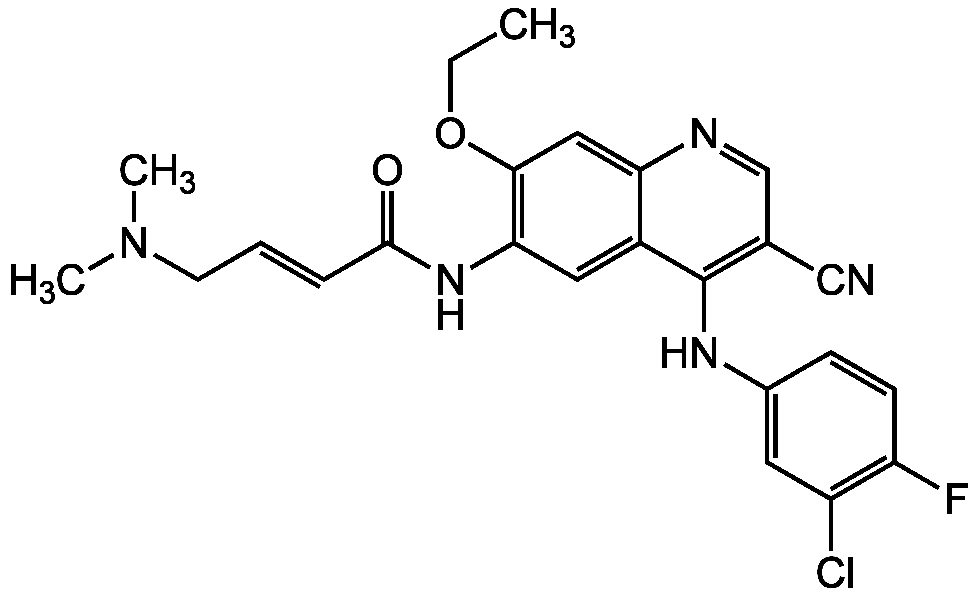
Pelitinib
| Code | Size | Price |
|---|
| SYN-1141-M100 | 100 mg | Enquire |
Quantity:
| SYN-1141-M001 | 1 mg | £104.00 |
Quantity:
| SYN-1141-M005 | 5 mg | £151.00 |
Quantity:
| SYN-1141-M010 | 10 mg | £256.00 |
Quantity:
| SYN-1141-M050 | 50 mg | £848.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
-20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
EKB-569
Appearance:
Solid.
CAS:
257933-82-7
EClass:
32160000
Form (Short):
liquid
GHS Symbol:
GHS07
Hazards:
H303, H317, H333
InChi:
InChI=1S/C24H23ClFN5O2/c1-4-33-22-12-20-17(11-21(22)30-23(32)6-5-9-31(2)3)24(15(13-27)14-28-20)29-16-7-8-19(26)18(25)10-16/h5-8,10-12,14H,4,9H2,1-3H3,(H,28,29)(H,30,32)/b6-5+
InChiKey:
WVUNYSQLFKLYNI-AATRIKPKSA-N
Long Description:
Chemical. CAS: 257933-82-7. Formula: C24H23ClFN5O2. MW: 467.9. Pelitinib, also known as EKB 569, is a potent, irreversible inhibitor of the EGFR tyrosine kinase. Cell based IC(50) values range from 39nM to 80nM and in vitro kinase assays are in sub-nanomolar range for EGFR receptors. In xenograft tumor models using overexpressing A431 cells Pelitinib inhibits growth of tumors with effective doses of 3.5-10mg/kg/dl. Recent research also shows that Pelitinib can potentiate radiation induced killing of squamouse cell carcinoma via the inhibition of IR-induced NF-kappaB mediated cell survial pathway.
Molecular Formula:
C24H23ClFN5O2
Molecular Weight:
467.9
Package Type:
Plastic Vial
Precautions:
P261, P272, P280, P302, P352, P333, P313, P321, P363, P501
Product Description:
Pelitinib, also known as EKB 569, is a potent, irreversible inhibitor of the EGFR tyrosine kinase. Cell based IC(50) values range from 39nM to 80nM and in vitro kinase assays are in sub-nanomolar range for EGFR receptors. In xenograft tumor models using overexpressing A431 cells Pelitinib inhibits growth of tumors with effective doses of 3.5-10mg/kg/dl. Recent research also shows that Pelitinib can potentiate radiation induced killing of squamouse cell carcinoma via the inhibition of IR-induced NF-kappaB mediated cell survial pathway.
Purity:
>95%
Signal word:
Warning
Solubility Chemicals:
Soluble in DMSO or ethanol.
Transportation:
Non-hazardous
UNSPSC Category:
Protein Kinase Modulators
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at -20°C.
References
Dual irreversible kinase inhibitors: quinazoline-based inhibitors incorporating two independent reactive centers with each targeting different cysteine residues in the kinase domains of EGFR and VEGFR-2: A. Wissner, et al.; Bioorg. Med. Chem. 15, 3635 (2007) | A quantitative analysis of kinase inhibitor selectivity: M.W. Karaman, et al.; Nat. Biotechnol. 26, 127 (2008) | Development of a new epidermal growth factor receptor positron emission tomography imaging agent based on the 3-cyanoquinoline core: synthesis and biological evaluation: F. Pisaneschi, et al.; Bioorg. Med. Chem. 18, 6634 (2010)
Related Products
| Product Name | Product Code | Supplier | Canertinib | SYN-1030 | AdipoGen Life Sciences | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gefitinib | SYN-1042 | AdipoGen Life Sciences | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lapatinib | SYN-1052 | AdipoGen Life Sciences | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afatinib | SYN-1100 | AdipoGen Life Sciences | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Foretinib | SYN-1129 | AdipoGen Life Sciences | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||