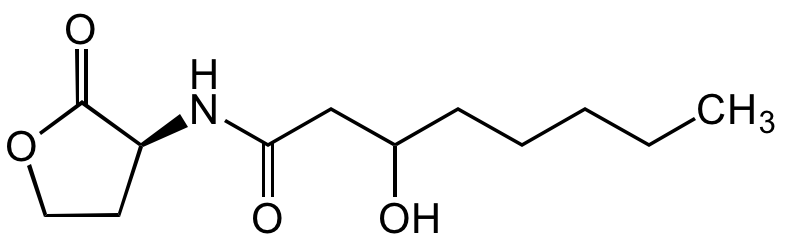
N-(3-Hydroxyoctanoyl)-L-homoserine lactone
| Code | Size | Price |
|---|
| CDX-H0206-M010 | 10 mg | £334.00 |
Quantity:
| CDX-H0206-M025 | 25 mg | £652.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
Ambient
Storage:
-20 °C
Images
Documents
Further Information
Alternate Names/Synonyms:
OH-C8-HSL; 3-Hydroxy-N-[(3S)-tetrahydro-2-oxo-3-furanyl]-octanamide
Appearance:
White to off-white powder.
CAS:
192883-14-0
EClass:
32160000
Form (Short):
liquid
Handling Advice:
Protect from light and moisture.
InChi:
InChI=1S/C12H21NO4/c1-2-3-4-5-9(14)8-11(15)13-10-6-7-17-12(10)16/h9-10,14H,2-8H2,1H3,(H,13,15)/t9?,10-/m0/s1
InChiKey:
XCZVBYOXRSFQBH-AXDSSHIGSA-N
Long Description:
Chemical. CAS: 192883-14-0. Formula: C12H21NO4. MW: 243.3. Synthetic. 3-Hydroxy-octanoyl-L-homoserine lactone is a small diffusible signaling molecule and is a member of N-acyl-homoserine lactone family. N-acylhomoserine lactones (AHL) are involved in quorum sensing, controlling gene expression, and cellular metabolism. The diverse applications of this kind of molecule include regulation of virulence in general, infection prevention, and formation of biofilms.
MDL:
MFCD31656747
Molecular Formula:
C12H21NO4
Molecular Weight:
243.3
Package Type:
Vial
Product Description:
3-Hydroxy-octanoyl-L-homoserine lactone is a small diffusible signaling molecule and is a member of N-acyl-homoserine lactone family. N-acylhomoserine lactones (AHL) are involved in quorum sensing, controlling gene expression, and cellular metabolism. The diverse applications of this kind of molecule include regulation of virulence in general, infection prevention, and formation of biofilms.
Purity:
>98% (HPLC)
SMILES:
O=C1[C@@H](NC(CC(O)CCCCC)=O)CCO1
Solubility Chemicals:
Soluble in DMSO, DMF or acetonitrile.
Source / Host:
Synthetic.
Transportation:
Non-hazardous
UNSPSC Category:
Biochemical Reagents
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at -20°C.
References
(1) L.R. Flodgaard, et al.; Appl. Environ. Microbiol. 71, 2113 (2005) | (2) P. Lumjiaktase, et al.; Microbiol. 152, 3651 (2006) | (3) B.A. Duerkop, et al.; J. Bacteriol. 190, 5137 (2008) | (4) A.M. Gamage, et al.; Microbiol. 157, 1176 (2011) | (5) K.G. Chan, et al.; BMC Microbiol. 11, 1 (2011)
Related Products
| Product Name | Product Code | Supplier | N-Hexadecanoyl-L-homoserine lactone | CDX-H0113 | Chemodex | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N-Hexanoyl-DL-homoserine lactone | CDX-H0200 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N-Heptanoyl-DL-homoserine lactone | CDX-H0201 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N-Hexadecanoyl-DL-homoserine lactone | CDX-H0202 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N-(3-Oxododecanoyl)-L-homoserine lactone | CDX-O0031 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N-(3-Oxobutanoyl)-L-homoserine lactone | CDX-O0037 | Chemodex | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||